3 In some ways, POC testing has more complex logistics and management in the hospital setting than tests performed on automated central laboratory platforms. 2 POC testing can potentially be performed in the hospital with a high level of quality and provide a unique and important role in patient care. Some laboratorians strongly feel that POC coagulation testing has a very limited role in a hospital setting (limited essentially to activated clotting time ), given the abilities of most central laboratories to provide a full menu of coagulation testing with rapid turnaround time (TAT). Nonwaived assays require liquid QC if an assay manufacturer recommends performing liquid QC less frequently than daily, Centers for Medicare & Medicaid Services may require an Individualized Quality Control Plan to ensure that the risks of less frequent QC performance are identified and mitigated. However, whether so-called electronic QC or true liquid QC is used differs by instrument and CLIA approval status. POC testing instruments typically have automatic system verification procedures. POC testing must be undertaken within the confines of safe practice involving adequate quality control procedures, competent testing staff (including well-trained self-testers), and use of POC assays solely in the patient population for which the POC assay was approved and valid. 2 The possibilities of preanalytic, analytic, and postanalytic errors are all potentially increased in the POC setting. POC testing can be plagued by insufficient calibration, quality control (QC), maintenance, and documentation. Regardless of the setting of POC use, testing is often performed by nonlaboratory personnel with less experience in quality control, in a less controlled environment than a typical clinical laboratory. Testing in the home requires US Food and Drug Administration (FDA) approval with waived status clinics and hospitals can use waived or moderate-complexity POC testing based on their staffing and accreditation. 
1ĭepending on the assay’s Clinical Laboratory Improvement Amendments (CLIA) status, POC testing can be performed in the hospital, clinic, or home. 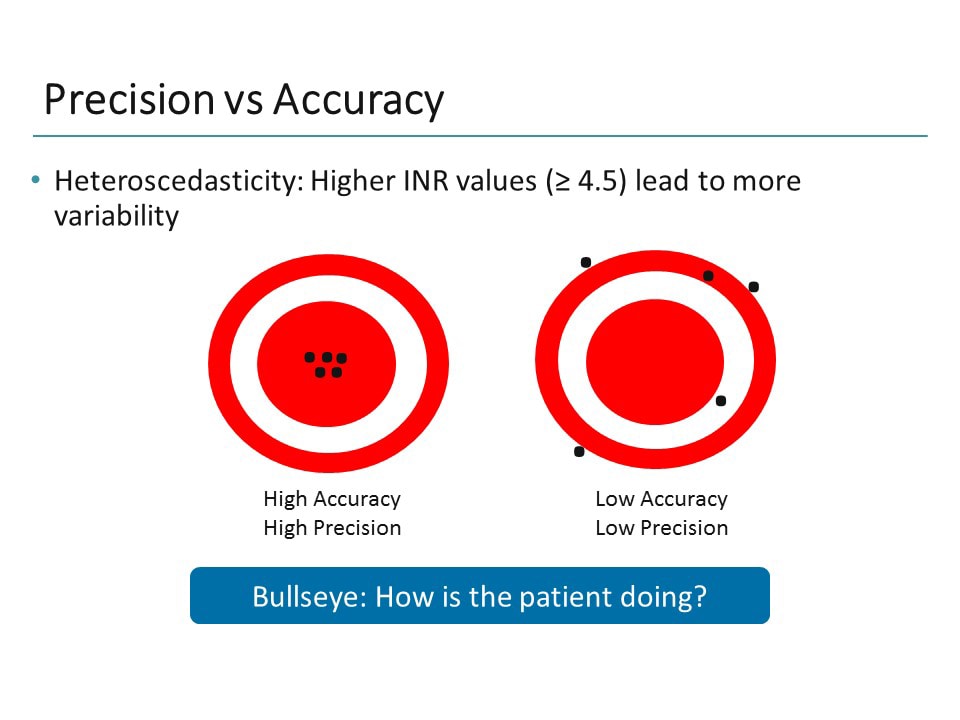
When considering POC testing, it is critically important to determine whether or not the imprecision and bias of POC testing are associated with clinically meaningful differences in test results relative to laboratory assays. This lesser performance as well as higher cost per test must be weighed against any clinical/logistical advantages of a POC device. In addition to increased imprecision, POC testing may also show bias relative to laboratory assays. Generally, the precision of POC systems is lower than that achieved with automated laboratory systems. These improvements are particularly helpful for patients in certain settings: intraoperative patients, patients with immediately life-threatening illness, and outpatients with chronic illnesses or medications that can be managed at home/nearby with the help of POC testing. POC testing can provide significant benefits in the form of improved time to test result/diagnosis or smaller specimen volume requirements. Point-of-care (POC) testing is performed at or near the site where clinical care is delivered, typically by personnel whose primary training is not in clinical laboratory science (to include patients performing self-testing).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
